|
Back to Blog
P periodic table5/17/2023 For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic tableĪs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.Phosphorus is also an essential ingredient of all cell protoplasm, nervous tissue, and bones. Trisodium phosphate is important as a cleaning agent, as a water softener, and for preventing boiler scale and corrosion of pipes and boiler tubes. Phosphorus is also important in the production of steels, phosphor bronze, and many other products. Phosphates are used in the production of special glasses, such as those used for sodium lamps.īone-ash -calcium phosphate- is used to create fine chinaware and to produce mono-calcium phosphate, used in baking powder. 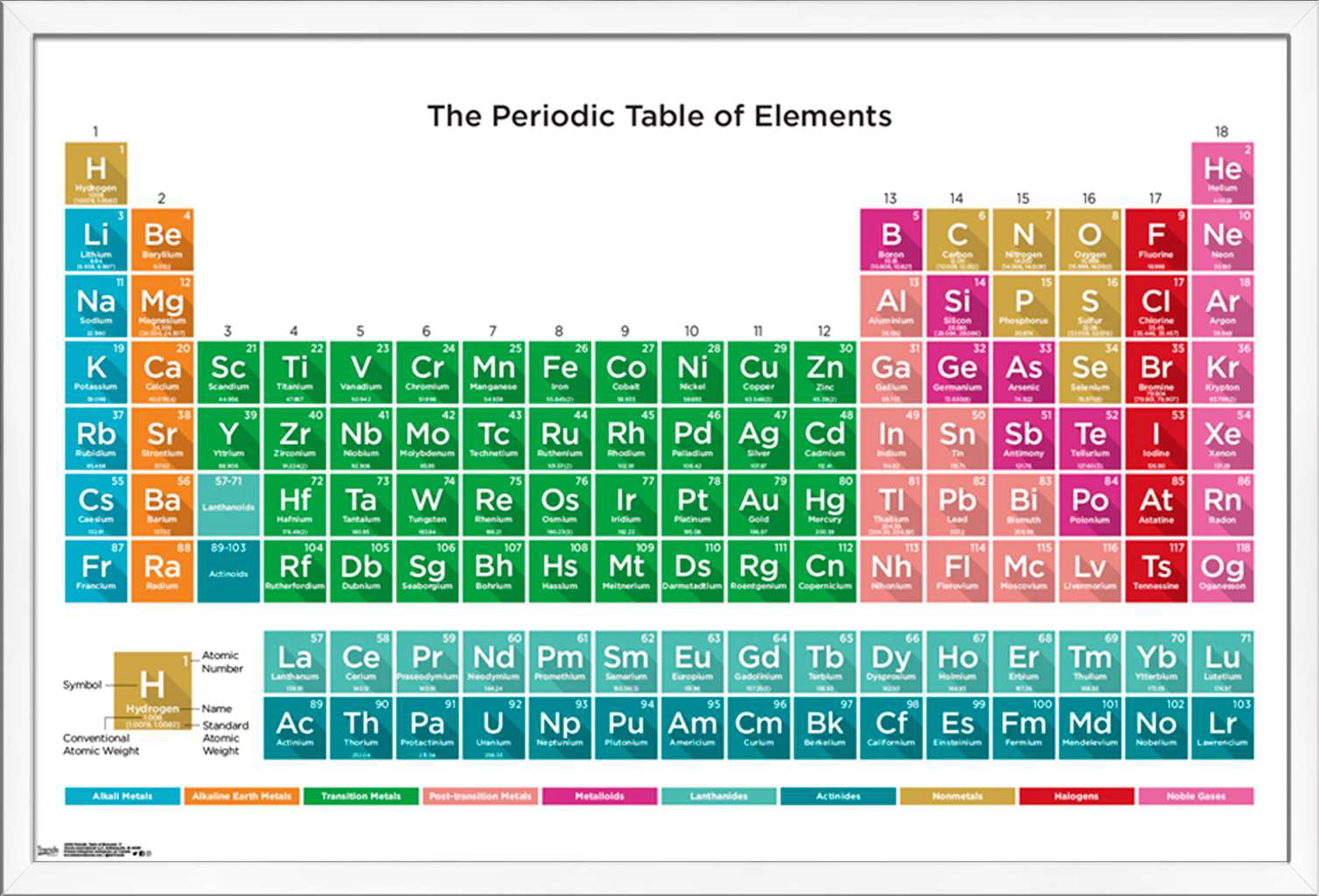
World-wide demand for fertilizers has caused record phosphate production. In recent years, concentrated phosphoric acids, which may contain as much as 70% to 75% P 2O 5 content, have become of great importance to agriculture and farm production. Elementary phosphorus is liberated as vapor and may be collected under phosphoric acid, an important compound in making super-phosphate fertilizers. By one process, tri-calcium phosphate, the essential ingredient of phosphate rock, is heated in the presence of carbon and silica in an electric furnace or fuel-fired furnace. 
White phosphorus may be made by several methods. 
The red modification is fairly stable, sublimes with a vapor pressure of 1 atm at 17C, and is used in the manufacture of safety matches, pyrotechnics, pesticides, incendiary shells, smoke bombs, tracer bullets, etc. It should, however, be handled with care as it does convert to the white form at some temperatures and it emits highly toxic fumes of the oxides of phosphorus when heated. This form does not ignite spontaneously and is not as dangerous as white phosphorus. When exposed to sunlight or when heated in its own vapor to 250☌, it is converted to the red variety, which does not phosphoresce in air as does the white variety. White phosphorus should be kept under water (as it is dangerously reactive in air) and should be handled with forceps, as contact with the skin may cause severe burns. Exposure to white phosphorus should not exceed 0.1 mg/m 3 (8-hour time-weighted average per 40-hour work week). 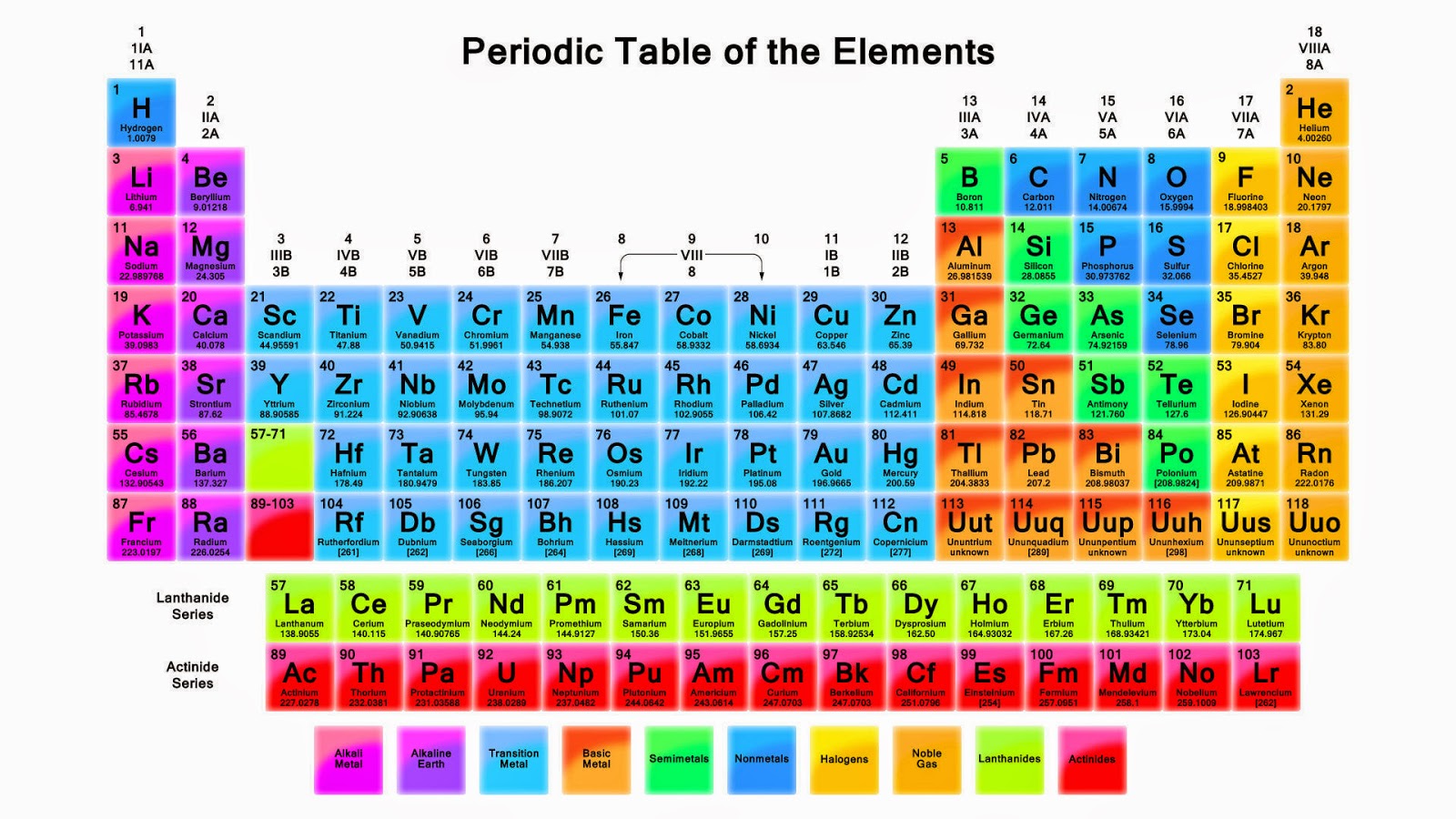
Phosphorus is very poisonous, 50 mg constituting an approximate fatal dose. Large deposits are found in Russia, in Morocco, and in Florida, Tennessee, Utah, Idaho, and elsewhere. Phosphate rock, which contains the mineral apatite, an impure tri-calcium phosphate, is an important source of the element. Never found free in nature, it is widely distributed in combination with minerals. It takes fire spontaneously in air, burning to the pentoxide. It is insoluble in water, but soluble in carbon disulfide. White phosphorus has two modifications: alpha and beta with a transition temperature at -3.8☌. Ordinary phosphorus is a waxy white solid when pure it is colorless and transparent. Phosphorus exists in four or more allotropic forms: white (or yellow), red, and black (or violet). Brand discovered phosphorus in 1669 by preparing it from urine. Phosphorus is a key ingredient in fertilizers (center) and the red in ordinary kitchen matches.ĥ, 4, 3, 2, 1, −1, −2, −3 (a mildly acidic oxide)įrom the Greek phosphoros, light bearing ancient name for the planet Venus when appearing before sunrise. Phosphates are used in the production of special glasses, such as those used for sodium lamps (street lights).
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
